Assessment |
Biopsychology |
Comparative |
Cognitive |
Developmental |
Language |
Individual differences |
Personality |
Philosophy |
Social |
Methods |
Statistics |
Clinical |
Educational |
Industrial |
Professional items |
World psychology |
Biological: Behavioural genetics · Evolutionary psychology · Neuroanatomy · Neurochemistry · Neuroendocrinology · Neuroscience · Psychoneuroimmunology · Physiological Psychology · Psychopharmacology (Index, Outline)
- Main article: Tomography
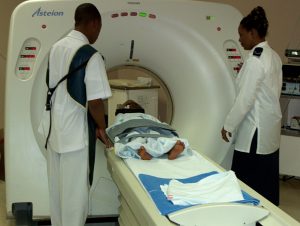
CT apparatus in a hospital
Computed tomography (CT), originally known as computed axial tomography (CAT or CT scan) and body section roentgenography, (and also known as computed axial tomography (CAT scan) X-ray computed tomography is a medical imaging method employing tomography where digital geometry processing is used to generate a three-dimensional image of the internals of an object from a large series of two-dimensional X-ray images taken around a single axis of rotation. The word "tomography" is derived from the Greek tomos (slice) and graphia (describing). CT produces a volume of data which can be manipulated, through a process known as windowing, in order to demonstrate various structures based on their ability to block the x-ray beam. Although historically (see below) the images generated were in the axial or transverse plane (orthogonal to the long axis of the body), modern scanners allow this volume of data to be reformatted in various planes or even as volumetric (3D) representations of structures.
Diagnostic use[]
Since its introduction in the 1970s, CT has become an important tool in medical imaging and neuroimaging to supplement X-rays and medical ultrasonography. Although it is still quite expensive, it is the gold standard in the diagnosis of a large number of different disease entities.
Cranial CT[]
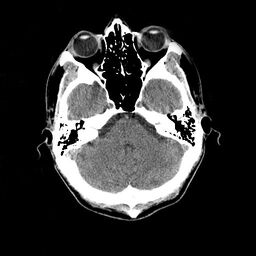
Normal CT scan of the head; this slice shows the cerebellum, a small portion of each temporal lobe, the orbits, and the ethmoid sinuses.
Diagnosis of cerebrovascular accidents and intracranial hemorrhage is the most frequent reason for a "head CT" or "CT brain". Scanning is done with or without intravenous contrast agents. CT generally does not exclude infarct in the acute stage of a stroke, but is useful to exclude a bleed (so anticoagulant medication can be commenced safely).
For detection of tumors, CT scanning with IV contrast is occasionally used but is less sensitive than magnetic resonance imaging (MRI).
CT can also be used to detect increases in intracranial pressure, e.g. before lumbar puncture or to evaluate the functioning of a ventriculoperitoneal shunt.
CT is also useful in the setting of trauma for evaluating facial and skull fractures.
In the head/neck/mouth area, CT scanning is used for surgical planning for craniofacial and dentofacial deformities, evaluation of cysts and some tumors of the jaws/paranasal sinuses/nasal cavity/orbits, diagnosis of the causes of chronic sinusitis, and for planning of dental implant reconstruction.
Chest CT[]
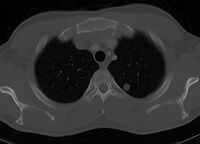
Chest CT horizontal section
CT is excellent for detecting both acute and chronic changes in the lung parenchyma. For detection of airspace disease (such as pneumonia) or cancer, ordinary non-contrast scans are adequate.
For evaluation of chronic interstitial processes (emphysema, fibrosis, and so forth), thin sections with high spatial frequency reconstructions are used. For evaluation of the mediastinum and hilar regions for lymphadenopathy, IV contrast is administered.
CT angiography of the chest (CTPA) is also becoming the primary method for detecting pulmonary embolism (PE) and aortic dissection, and requires accurately timed rapid injections of contrast and high-speed helical scanners. CT is the standard method of evaluating abnormalities seen on chest X-ray and of following findings of uncertain acute significance.
Cardiac CT[]
With the advent of subsecond rotation combined with multi-slice CT (up to 64 slices), high resolution and high speed can be obtained at the same time, allowing excellent imaging of the coronary arteries. Images with a high temporal resolution are formed by updating a proportion of the data set used for image reconstruction as it is scanned. In this way individual frames in a cardiac CT investigation are significantly shorter than the shortest tube rotation time. It is uncertain whether this modality will replace the invasive coronary catheterization.
Cardiac MSCT carries very real risks since it exposes the subject to the equivalent of 500 chest X Rays in terms of radiation. The relationship of radiation exposure to increased risk in breast cancer has yet to be definitively explored.
The positive predictive value is approximately 82% while the negative predictive value is in the range of 93%. Sensitivity is about 81% and the specificity is about 94%. The real benefit in the test is the high negative predictive value. Thus, when the coronary arteries are free of disease by CT, patients can then be worked up for other causes of chest symptoms.
Much of the software is based on data findings from caucasian study groups and as such the assumptions made may also not be totally true for all other populations.
Dual Source CT scanners, introduced in 2005, allow higher temporal resolution so reduce motion blurring at high heart rates, and potentially requiring a shorter breath-hold time. This is particularly useful for ill patient who have difficult holding their breath, or who are unable to take heart-rate lowering medication.
Abdominal and pelvic CT[]
CT is a sensitive method for diagnosis of abdominal diseases. It is used frequently to determine stage of cancer and to follow progress. It is also a useful test to investigate acute abdominal pain. Renal/urinary stones, appendicitis, pancreatitis, diverticulitis, abdominal aortic aneurysm, and bowel obstruction are conditions that are readily diagnosed and assessed with CT. CT is also the first line for detecting solid organ injury after trauma.
Oral and/or rectal contrast may be used depending on the indications for the scan. A dilute (2% w/v) suspension of barium sulfate is most commonly used. The concentrated barium sulfate preparations used for fluoroscopy e.g. barium enema are too dense and cause severe artifacts on CT. Iodinated contrast agents may be used if barium is contraindicated (e.g. suspicion of bowel injury). Other agents may be required to optimize the imaging of specific organs: e.g. rectally administered gas (air or carbon dioxide) for a colon study, or oral water for a stomach study.
CT has limited application in the evaluation of the pelvis. For the female pelvis in particular, ultrasound is the imaging modality of choice. Nevertheless, it may be part of abdominal scanning (e.g. for tumors), and has uses in assessing fractures.
CT is also used in osteoporosis studies and research along side DXA scanning. Both CT and DXA can be used to asses bone mineral density (BMD) which is used to indicate bone strength, however CT results do not correlate exactly with DXA (the gold standard of BMD measurment). DXA is far more expensive, and subjects patients to much higher levels of ionizing radiation, so it is used infrequently.
Advantages and hazards[]
Advantages Over Projection Radiography (See Radiography)[]
First, CT completely eliminates the superimposition of images of structures outside the area of interest. Second, because of the inherent high-contrast resolution of CT, differences between tissues that differ in physical density by less than 1% can be distinguished. Third, data from a single CT imaging procedure consisting of either multiple contiguous or one helical scan can be viewed as images in the axial, coronal, or sagittal planes, depending on the diagnostic task. This is referred to as multiplanar reformatted imaging.
Radiation exposure[]
CT is regarded as a moderate to high radiation diagnostic technique. While technical advances have improved radiation efficiency, there has been simultaneous pressure to obtain higher-resolution imaging and use more complex scan techniques, both of which require higher doses of radiation. The improved resolution of CT has permitted the development of new investigations, which may have advantages; e.g. Compared to conventional angiography, CT angiography avoids the invasive insertion of an arterial catheter and guidewire; CT colonography may be as good as barium enema for detection of tumors, but may use a lower radiation dose.
The greatly increased availability of CT, together with its value for an increasing number of conditions, has been responsible for a large rise in popularity. So large has been this rise that, in the most recent comprehensive survey in the UK, CT scans constituted 7% of all radiologic examinations, but contributed 47% of the total collective dose from medical X-ray examinations in 2000/2001 (Hart & Wall, European Journal of Radiology 2004;50:285-291). Increased CT usage has led to an overall rise in the total amount of medical radiation used, despite reductions in other areas.
The radiation dose for a particular study depends on multiple factors: volume scanned, patient build, number and type of scan sequences, and desired resolution and image quality.
Typical scan doses[]
| Examination | Typical effective dose (mSv) |
|---|---|
| Chest X-ray | 0.02 |
| Head CT | 1.5(a) |
| Abdomen | 5.3(a) |
| Chest | 5.8(a) |
| Chest, Abdomen and Pelvis | 9.9(a) |
| Cardiac CT angiogram | 6.7-13(b) |
| CT colongraphy (virtual colonoscopy) | 3.6 - 8.8 |
Adverse reactions to contrast agents[]
Because CT scans rely on intravenously administered contrast agents in order to provide superior image quality, there is a low but non-negligible level of risk associated with the contrast agents themselves. Certain patients may experience severe and potentially life-threatening allergic reactions to the contrast dye.
The contrast agent may also induce kidney damage. The risk of this is increased with patients who have preexisting renal insufficiency, preexisting diabetes, or reduced intravascular volume. In general, if a patient has normal kidney function, then the risks of contrast nephropathy are negligable. Patients with mild kidney impairment are usually advised to ensure full hydration for several hours before and after the injection. For moderate kidney failure, the use of iodinated contrast should be avoided; this may mean using an alternative technique instead of CT e.g. MRI. Perhaps paradoxically, patients with severe renal failure requiring dialysis do not require special precautions, as their kidneys have so little function remaining that any further damage would not be noticable and the dialysis will remove the contrast agent.
Process[]
X-ray slice data is generated using an X-ray source that rotates around the object; X-ray sensors are positioned on the opposite side of the circle from the X-ray source. Many data scans are progressively taken as the object is gradually passed through the gantry. They are combined together by the mathematical procedure known as tomographic reconstruction.
Newer machines with faster computer systems and newer software strategies can process not only individual cross sections but continuously changing cross sections as the gantry, with the object to be imaged, is slowly and smoothly slid through the X-ray circle. These are called helical or spiral CT machines. Their computer systems integrate the data of the moving individual slices to generate three dimensional volumetric information (3D-CT scan), in turn viewable from multiple different perspectives on attached CT workstation monitors.

CT scanner with cover removed to show the principle of operation
In conventional CT machines, an X-Ray tube and detector are physically rotated behind a circular shroud (see the image above right); in the electron beam tomography (EBT) the tube is far larger and higher power to support the high temporal resolution. The electron beam is deflected in a hollow funnel shaped vacuum chamber. Xray is generated when the beam hits a stationary target. The detector is also stationary.
The data stream representing the varying radiographic intensity sensed reaching the detectors on the opposite side of the circle during each sweep is then computer processed to calculate cross-sectional estimations of the radiographic density, expressed in Hounsfield units. Sweeps cover 360 or just over 180 degrees in conventional machines, 220 degrees in EBT.
CT is used in medicine as a diagnostic tool and as a guide for interventional procedures. Sometimes contrast materials such as intravenous iodinated contrast are used. This is useful to highlight structures such as blood vessels that otherwise would be difficult to delineate from their surroundings. Using contrast material can also help to obtain functional information about tissues.
Pixels in an image obtained by CT scanning are displayed in terms of relative radiodensity. The pixel itself is displayed according to the mean attenuation of the tissue(s) that it corresponds to on a scale from -1024 to +3071 on the Hounsfield scale. Pixel is a two dimensional unit based on the matrix size and the field of view. When the CT slice thickness is also factored in, the unit is known as a Voxel, which is a three dimensional unit. The phenomenon that one part of the detector can not differ between different tissues is called the Partial Volume Effect. That means that a big amount of cartilage and a thin layer of compact bone can cause the same attenuation in a voxel as hyperdense cartilage alone. Water has an attenuation of 0 Hounsfield units (HU) while air is -1000 HU, cancellous bone is typically +400 HU, cranial bone can reach 2000 HU or more (os temporale) and can cause artefacts. The attenuation of metallic implants depends on atomic number of the element used: Titanium usually has an amount of +1000 HU, iron steel can completely extinguish the X-ray and is therefore responsible for well-known line-artefacts in computed tomogrammes.
Windowing[]
Windowing is the process of using the calculated Hounsfield units to make an image. The various radiodensity amplitudes are mapped to 256 shades of gray. These shades of gray can be distributed over a wide range of HU values to get an overview of structures that attenuate the beam to widely varying degrees. Alternatively, these shades of gray can be distributed over a narrow range of HU values (called a narrow window) centered over the average HU value of a particular structure to be evaluated. In this way, subtle variations in the internal makeup of the structure can be discerned. This is a commonly used image processing technique known as contrast compression. For example, to evaluate the abdomen in order to find subtle masses in the liver, one might use liver windows. Choosing 70 HU as an average HU value for liver, the shades of gray can be distributed over a narrow window or range. One could use 170 HU as the narrow window, with 85 HU above the 70 HU average value; 85 HU below it. Therefore the liver window would extend from -15 HU to +155 HU. All the shades of gray for the image would be distributed in this range of Hounsfield values. Any HU value below -15 would be pure black, and any HU value above 155 HU would be pure white in this example. Using this same logic, bone windows would use a wide window (to evaluate everything from fat-containing medullary bone that contains the marrow, to the dense cortical bone), and the center or level would be a value in the hundreds of Hounsfield units.
Three dimensional (3D) reconstruction[]
The principle[]
Because contemporary CT scanners offer isotropic, or near isotropic, resolution, display of images does not need to be restricted to the conventional axial images. Instead, it is possible for a software program to build a volume by 'stacking' the individual slices one on top of the other. The program may then display the volume in an alternative manner.
Multiplanar reconstruction[]
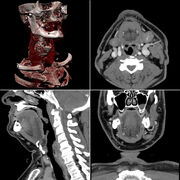
Typical screen layout for diagnostic software, showing 1 3D and 3 MPR views
This is the simplest method of reconstruction. A volume is built by stacking the axial slices. The software then cuts slices through the volume in a different plane (usually orthogonal). Optionally, a special projection method (maximum-intensity projection (MIP) or minimum-intensity projection (mIP) can be used to build the reconstructed slices.
MPR is frequently used for examining the spine. Axial images through the spine will only show one vertebral body at a time and cannot reliably show the intervertebral discs. By reformatting the volume, it becomes much easier to visualise the position of one vertebral body in relation to the others.
Modern software allows reconstruction in non-orthogonal (oblique) planes so that the optimal plane can be chosen to display an anatomical structure. This may be particularly useful for visualising the structure of the bronchi as these do not lie orthogonal to the direction of the scan.
For vascular imaging, curved-plane reconstruction can be performed. This allows bends in a vessel to be 'straightened' so that the entire length can be visualised on one image, or a short series of images. Once a vessel has been 'straightened' in this way, quantitative measurements of length and cross sectional area can be made, so that surgery or interventional treatment can be planned.
MIP reconstructions enhance areas of high radiodensity, and so are useful for angiographic studies. mIP reconstructions tend to enhance air spaces so are useful for assessing lung structure.
3D rendering techniques[]
Surface rendering: A threshold value of radiodensity is chosen by the operator (e.g. a level that corresponds to bone). A threshold level is set, using edge detection image processing algorithms. From this, a 3-dimensional model can be constructed and displayed on screen. Multiple models can be constructed from various different thresholds, allowing different colors to represent each anatomical component such as bone, muscle, and cartilage. However, the interior structure of each element is not visible in this mode of operation.
Volume rendering: Surface rendering is limited in that it will only display surfaces which meet a threshold density, and will only display the surface that is closest to the imaginary viewer. In volume rendering, transparency and colors are used to allow a better representation of the volume to be shown in a single image - e.g. the bones of the pelvis could be displayed as semi-transparent, so that even at an oblique angle, one part of the image does not conceal another.
Image segmentation[]
Where different structures have similar radiodensity, it can become impossible to separate them simply by adjusting volume rendering parameters. The solution is called segmentation, a manual or automatic procedure that can remove the unwanted structures from the image.
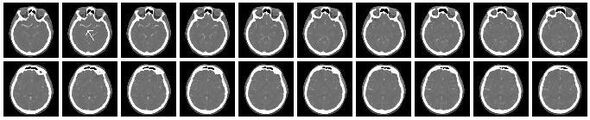
Example[]
Some slices of a cranial CT scan are shown below. The bones are whiter than the surrounding area. (Whiter means higher radiodensity.) Note the blood vessels (arrowed) showing brightly due to the injection of an iodine-based constrast agent.
A volume rendering of this volume clearly shows the high density bones.
After using a segmentation tool to remove the bone, the previously concealed vessels can now be demonstrated.
History[]
The first commercially viable CT system was invented by Godfrey Newbold Hounsfield in Hayes, England at THORN EMI Central Research Laboratories using X-rays. Hounsfield conceived his idea in 1967, and it was publicly announced in 1972. It is claimed that the CT scanner was "the greatest legacy" of the Beatles; the massive profits from their record sales enabled EMI to fund scientific research [3]. Allan McLeod Cormack of Tufts University independently invented a similar process at the University of Cape Town/Groote Schuur Hospital and they shared a Nobel Prize in medicine in 1979.

The prototype CT scanner
The original 1971 prototype took 160 parallel readings through 180 angles, each 1° apart, with each scan taking a little over five minutes. The images from these scans took 2.5 hours to be processed by algebraic reconstruction techniques on a large computer.
The first production X-ray CT machine (called the EMI-Scanner) was limited to making tomographic sections of the brain, but acquired the image data in about 4 minutes (scanning two adjacent slices) and the computation time (using a Data General Nova minicomputer) was about 7 minutes per picture. This scanner required the use of a water-filled Perspex tank with a pre-shaped rubber "head-cap" at the front, which enclosed the patient's head. The water-tank was used to reduce the dynamic range of the radiation reaching the detectors (between scanning outside the head compared with scanning through the bone of the skull). The images were relatively low resolution, being composed of a matrix of only 80 x 80 pixels. The first EMI-Scanner was installed in Atkinson Morley's Hospital in Wimbledon, England, and the first patient brain-scan was made with it in 1972.

an historic EMI-Scanner
In the US, the machine sold for about $390,000, with the first installations being at the Lahey Clinic, then Massachusetts General Hospital, and George Washington University in 1973.
The first CT system that could make images of any part of the body, and did not require the "water tank" was the ACTA scanner designed by Robert S. Ledley, DDS at Georgetown University.
CT technology generations[]
- First generation: These CT scanners used a pencil-thin beam of radiation directed at one or two detectors. The images were acquired by a "translate-rotate" method in which the x-ray source and the detector in a fixed relative position move across the patient followed by a rotation of the x-ray source/detector combination (gantry) by one degree. In the EMI-Scanner, a pair of images was acquired in about 4 minutes with the gantry rotating a total of 180 degrees. Three detectors were used (one of these being an X-ray source reference), each detector comprising a sodium iodide scintillator and a photomultiplier tube. Some patients had unpleasant experiences within these early scanners, due to the loud sounds and vibrations from the equipment.
- Second generation: This design increased the number of detectors and changed the shape of the radiation beam. The x-ray source changed from the pencil-thin beam to a fan shaped beam. The "translate-rotate" method was still used but there was a significant decrease in scanning time. Rotation was increased from one degree to thirty degrees.
- Third generation: CT scanners made a dramatic change in the speed at which images could be obtained. In the third generation a fan shaped beam of x-rays is directed to an array of detectors that are fixed in position relative to the x-ray source. This eliminated the time consuming translation stage allowing scan time to be reduced, initially, to 10 seconds per slice. This advance dramatically improved the practicality of CT. Scan times became short enough to image the lungs or the abdomen; previous generations had been limited to the head, or to limbs. Patients have reported more pleasant experiences with the third and fourth generation CT scanners because of greatly reduced noise and vibration compared to earlier models.
- Fourth generation: This design was introduced, roughly simultaneously with 3rd generation, and gave approximately equal performance. Instead of a row of detectors which moved with the X-ray source, 4th generation scanners used a stationary 360 degree ring of detectors. The fan shaped x-ray beam rotated around the patient directed at detectors in a non-fixed relationship.
Bulky, expensive and fragile photomultiplier tubes gradually gave way to improved detectors. A xenon gas ionization chamber detector array was developed for third generation scanners, which provided greater resolution and sensitivity. Eventually, both of these technologies were replaced with solid-state detectors: rectangular, solid-state photodiodes, coated with a fluorescent rare earth phosphor. Solid state detectors were smaller, more sensitive and more stable, and were suitable for 3rd and 4th generation designs.
On an early 4th generation scanner, 600 photomultiplier tubes, ½ in. (12 mm) in diameter, could fit in the detector ring. Three photodiode units could replace one photomultiplier tube. This change resulted in increasing both the acquisition speed, and image resolution. The method of scanning was still slow, because the X-ray tube and control components interfaced by cable, limiting the scan frame rotation.
Initially, 4th generation scanners carried a significant advantage - the detectors could be automatically calibrated on every scan. The fixed geometry of 3rd generation scanners was especially sensitive to detector mis-calibration (causing ring artifacts). Additionally, because the detectors were subject to movement and vibration, their calibration could drift significantly.
All modern medical scanners are of 3rd generation design. Modern solid-state detectors are sufficiently stable that calibration for each image is no longer required. The 4th generation scanners' inefficient use of detectors made them considerably more expensive than 3rd generation scanners. Further, they were more sensitive to artifacts because the non-fixed relationship to the x-ray source made it impossible to reject scattered radiation.
Further advances[]
Another limiting factor in image acquisition was the X-ray tube. The need for long, high intensity exposures and very stable output placed enormous demands on both the tube and generator (power supply). Very high performance rotating anode tubes were developed to keep up with demand for faster imaging, as were the regulated 150 kV switched mode power supplies to drive them. Modern systems have power ratings up to 100 kW.
Slip-ring technology replaced the spooled cable technology of older CT scanners, allowing the X-ray tube and detectors to spin continuously. When combined with the ability to move the patient continuously through the scanner this refinement is called Helical CT or, more commonly, Spiral CT.
Multi-detector-row CT systems further accelerated scans, by allowing several images to be acquired simultaneously. Modern scanners are available with up to 64 detector rows / output channels ( depends upon the technology used by the manufacturer ). It is possible to complete a scan of the chest in a few seconds. An examination that required 10 separate breath-holds of 10 seconds each can now be completed in a single 10 second breath-hold. Multi-detector CT can also provide isotropic resolution, permitting cross-sectional images to be reconstructed in arbitrary planes; an ability similar to MRI. More anatomical volume coverage in less time is one of the key features of the latest generation MD CT Scanners. It is however more important to achieve better spatial resolution than only volume coverage for better reconstructed images. Latest generation MD CT scanners with flying X-Ray tube focal spot in z-axis direction shows better image resolution. A different approach was used for a particular type of dedicated cardiac CT technique called electron-beam CT (also known as ultrafast CT, and occasionally fifth generation CT). With temporal resolution of approximately 50 ms, these scanners could freeze cardiac and pulmonary motion providing high quality images. Only one manufacturer offered these scanners (Imatron, later GE healthcare), and few of these scanners were ever installed, primarily due to the very high cost of the equipment and their single-purpose design. Rapid development of MDCT has significantly reduced the advantage of EBCT over conventional systems. Contemporary MDCT systems have temporal resolution approaching that of EBCT, but at lower cost and with much higher flexibility. Because of this, MDCT is usually the preferred choice for new installations.
Improved computer technology and reconstruction algorithms have permitted faster and more accurate reconstruction. On early scanners reconstruction could take several minutes per image, a modern scanner can reconstruct a 1000 image study in under 30 seconds. Refinements to the algorithms have reduced artifacts. Dual source CT uses 2 x-ray sources and 2 detector arrays offset at 90 degrees. This reduces the time to acquire each image to about 0.1 seconds, making it possible to obtain high quality images of the heart without the need for heart rate lowering drugs such as beta blockers. A dual-source multi-detector row scanner can complete an entire cardiac study within a single 10 second breath hold.
Volumetric CT is an extension of multi-detector CT, currently at research stage. Current MDCT scanners sample a 4 cm wide volume in one rotation. Volumetric CT aims to increase the scan width to 10-20 cm, with current prototypes using 256 detector-rows. Potential applications include cardiac imaging (a complete 3D dataset could be acquired in the time between 2 successive beats) and 3D cine-angiography.
Microtomography[]
In recent years, tomography has also been introduced on the micrometer level and is named Microtomography. But these machines are currently only fit for smaller objects or animals, and cannot yet be used on humans.
See also[]
- Cardiology diagnostic tests and procedures
- Computed Tomography Laser Mammography (CTLM)
- Fluoroscopy
- Medical ultrasonography
- Magnetic resonance imaging (MRI)
- Neuroimaging
- Positron emission tomography (PET)
- Single photon emission computed tomography (SPECT)
- Electron-beam computed tomography (EBCT)
- Digitally Reconstructed Radiograph
- InVesalius
- Synchrotron X-ray tomographic microscopy
References & Bibliography[]
Key texts[]
Books[]
Papers[]
Additional material[]
Books[]
Papers[]
External links[]
- CTisus comprehensive CT site by Dr. Elliot Fishman of Johns Hopkins University
- MultiSlice CT Angiogram from Angioplasty.Org
- RadiologyInfo- The radiology information resource for patients: Computed Tomography
- Example CT Scan
| This page uses Creative Commons Licensed content from Wikipedia (view authors). |