No edit summary |
|||
| Line 182: | Line 182: | ||
== See also == |
== See also == |
||
| + | * [[Conditioned suppression]] |
||
| − | |||
* [[Latent inhibition]] |
* [[Latent inhibition]] |
||
* [[P50 evoked potential]] – a measure of sensory processing related to PPI but uncorrelated to it. P50 and PPI could be measured in a combined single session, providing complementary data about sensory and sensorymotor gating.<!-- |
* [[P50 evoked potential]] – a measure of sensory processing related to PPI but uncorrelated to it. P50 and PPI could be measured in a combined single session, providing complementary data about sensory and sensorymotor gating.<!-- |
||
| + | * [[Sensory gating]] |
||
| − | |||
| + | * [[Startle reflex]] |
||
--><ref name=”Light_2001_combined_P50_PPI>Light GA, Braff DL. (2001) ''Measuring P50 suppression and prepulse inhibition in a single recording session.'' Am J Psychiatry. 2001 Dec;158(12):2066-8. PMID 11729028 [http://ajp.psychiatryonline.org/cgi/content/full/158/12/2066 free full text] </ref><!-- |
--><ref name=”Light_2001_combined_P50_PPI>Light GA, Braff DL. (2001) ''Measuring P50 suppression and prepulse inhibition in a single recording session.'' Am J Psychiatry. 2001 Dec;158(12):2066-8. PMID 11729028 [http://ajp.psychiatryonline.org/cgi/content/full/158/12/2066 free full text] </ref><!-- |
||
Revision as of 22:08, 11 January 2010
Assessment |
Biopsychology |
Comparative |
Cognitive |
Developmental |
Language |
Individual differences |
Personality |
Philosophy |
Social |
Methods |
Statistics |
Clinical |
Educational |
Industrial |
Professional items |
World psychology |
Animals · Animal ethology · Comparative psychology · Animal models · Outline · Index
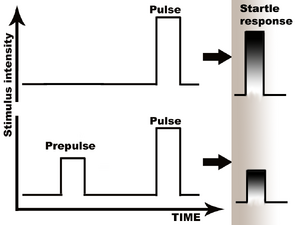
Prepulse inhibition: preceding stimulus attenuates the startle response.
Prepulse Inhibition (PPI) is a neurological phenomenon in which a weaker prestimulus (prepulse) inhibits the reaction of an organism to a subsequent strong startling stumulus (pulse). The stimuli are usually acoustic, but tactile, light, airpuff stimuli are also used. The reduction of the amplitude of startle reflects the ability of the nervous system to temporarily adapt to a strong sensory stimulus when a preceding weaker signal is given to warn the organism. PPI is detected in numerous species ranging from mice to human. Although the extent of the adaptation affects numerous systems, the most comfortable to measure are the muscular reactions, which are normally diminished as a result of the nervous inhibition.
Deficits of prepulse inhibition manifest in the inability to filter out the unnecessary information; they have been linked to abnormalities of sensorimotor gating. Such deficits are noted in patients suffering from illnesses like schizophrenia and Alzheimer’s disease, and in people under the influence of drugs, surgical manipulations, mutations. Human studies of PPI have been summarised in a review by Braff et. al. in 2001.[1]
Procedure
The main three parts of the procedure are prepulse, startle stimulus, and startle reflex. Different prepulse-to-pulse intervals, or lead intervals, are used: 30, 60, 120, 240 and 480 ms. Lead interval counts from the start of prepulse to the start of the pulse. With the interval exceeding 500 ms, prepulse facilitation - increased response - is most likely to follow. Burst of white noise is usually used as acoustic startle stimulus. Typical durations are 20 ms for prepulse and 40 ms for pulse. Background noise with 65-70 dB is used in human studies, and 30-40 dB in rodent experiments. Prepulse is typically set 3-12 dB louder than background. Startle response is measured in rodents using the so-called automated “startle chambers” or “stabilimeter chambers”, with detectors recording whole-body reaction. In humans, the movements of oculomotor muscles (“eye-blink reflex” or “eye-blink response” assessed using electromyographic recording of orbicularis oculi muscle and by oculography) could be used as a measure. Pulse-alone results are compared to prepulse-plus-pulse, and the percentage of the reduction in the startle reflex represents prepulse inhibition. Possible hearing impairment must be taken into account, as, for example, several strains of mice develop high frequency hearing loss when they mature.[2]
Major features
- The magnitude of PPI is often significant, reaching as much as 65% in healthy subjects.
- Maximum inhibition is typically observed at 120 ms interval.[3]
- Baseline startle response does not affect overall PPI levels – this finding was first discovered in rat studies[4] and later duplicated in the studies of mice.[5]
- The opposite reaction, Prepulse Facilitation (PPF), is typically noted when the interval between stimuli lasts longer than 500 ms. PPF is thought to reflect, at least partially, sustained attention.
- There is noted sex difference in prepulse inhibition, with men having higher PPI, while women having higher PPF.[6]
- Even the very first prepulse of the test session induces inhibition, which indicates that conditioning and learning are not necessary for this effect to occur. However, the lack of conditionality has been questioned.[9]
- It is thought that the short intervals used in PPI task do not give enough time for the activation of a volitional response.
- Prepulses could be attended or ignored, and attention affects the outcome. In one study, normal college students were instructed to attend to one of the kind of prepulses, high- or low-pitched, and ignore the other. Attended prepulse caused significantly greater inhibition at the 120 ms interval compared to the ignored one, and significantly greater facilitation at the 2000 ms interval.[10]
- Louder background noise increases the amplitude of the startle response.
- Increased prepulse duration leads to increase in PPI.
- Steady background noise facilitates the startle response, while pulsed background produces inhibition.[11]
Disruption of PPI
Disruptions of PPI are studied in humans and many other species. The most studied are deficits of PPI in schizophrenia, although this disease is not the only one to cause such deficits. They have been noted in schizotypal personality disorder,[12] obsessive-compulsive disorder(Swerdlow et. al., 1993), Huntington's disease,[13] nocturnal enuresis and attention deficit disorder (Ornitz et al. 1992), and Tourette's syndrome (Swerdlow et al. 1994; Castellanos et al. 1996). According to one study, people who have temporal lobe epilepsy with psychosis also show decreases in PPI, unlike those who have TLE without psychosis.[14] Therefore, PPI deficits are not typical to specific disease, but rather tell of disruptions in a specific brain circuit.
PPI deficit in schizophrenia
PPI deficits represent a well-described finding in schizophrenia, with the first report dating back to 1978 [15] The abnormalities are also noted in unaffected relatives of the patients.[16][17] In one study, patients failed to show increased PPI to attended prepulses.[18] Dopamine, which plays a major role in schizophrenia, had been shown to regulate sensorimotor gating in rodent models.[19][20] These findings fit to the dopamine hypothesis of schizophrenia. In theory, PPI disruption in schizophrenia may be related to the processes of sensory flooding and cognitive fragmentation.
Antipsychotic medication have been shown to increase PPI in patients, with atypical antipsychotics having more effect. Patients display the same gender difference in PPI as healthy people: males have higher PPI compared to females. One notable finding is that patients are specifically deficient in PPI with 60 ms prepulse intervals relative to intervals of other lengths; this remains so even under antipsychotic treatment.[21]
The other fact is the influence of cigarette smoking. Non-smoking patients have lower PPI compared to smokers, and heavy smokers have the highest PPI.[21][22] This finding runs in accord with the high rates of smoking among schizophrenic patients, estimated at 70%,[23] with many patients smoking more than 30 cigarettes a day.[24] Thus, smoking may be a way of self-medication. Some studies show association of schizophrenia with the CHRNA7 and CHRFAM7A genes, which code for alpha7 subunit of nicotinic receptors, but other studies are negative.[25][26] Contrary to the predictions, nicotine receptor alpha7 subunit knockout mice do not show disruptions in PPI.[27]
Disruption of PPI in rodents
Murine models are widely used to test hypotheses linking genetic components of various diseases with sensorimotor gating. While some of the hypotheses stand to the test, others are not, as some mice models show unchanged or increased PPI contrary to the expectations, as in the tests of COMT-deficient mice.[28]
Certain surgical procedures also disrupt PPI in animals, helping to unravel the underlying circuitry.
Many animal studies of PPI are undertaken in order to understand and model the pathology of schizophrenia.[29] Schizophrenia-like PPI disruption techniques in rodents have been classified in one review [30] into four models:
- PPI impairment driven by dopamine-receptor agonists, most validated for antipsychotic studies;
- PPI impairment by 5-HT2 receptor agonists;
- PPI impairment by NMDAR antagonists;
- PPI impairment by developmental intervention (isolation rearing, maternal deprivation).
Diverse chemical compounds are tested on animals with such deficits. Compounds that are able to restore PPI could be further investigated for their potential antipsychotic role.
See also
- Conditioned suppression
- Latent inhibition
- P50 evoked potential – a measure of sensory processing related to PPI but uncorrelated to it. P50 and PPI could be measured in a combined single session, providing complementary data about sensory and sensorymotor gating.[31]
External links
- Table listing the action of different chemical compounds on PPI in mice - from the review by Geyer et. al., 2002. [2]
- Losing Your (Prepulse) Inhibitions—All About α3 GABAA? - Schizophrenia Research Forum.
- Prepulse Inhibition Deficits Predict Functional Difficulties in Schizophrenia - Schizophrenia Research Forum.
- An Analysis of Nicotine Exacerbation of Reductions in PPI in a Rodent Model of Schizophrenia - a master's thesis with a thorough review of schizophrenia PPI disruption models in rodents.
References
- ↑ Braff, DL, Geyer, MA, Swerdlow, NR. Human studies of prepulse inhibition of startle: normal subjects, patient groups, and pharmacological studies. Psychopharmacology 2001; 156:234–258 PMID 11549226
- ↑ 2.0 2.1 Geyer MA, McIlwain KL, Paylor R. (2002) Mouse genetic models for prepulse inhibition: an early review. Mol Psychiatry. 2002;7(10):1039-53. PMID 12476318 free fulltext
- ↑ Graham FK (1975). The more or less startling effects of weak prestimulation. Psychophysiology 12: 238-248. PMID 1153628
- ↑ Swerdlow NR, Geyer MA, Braff DL. (2001) Neural circuitry of prepulse inhibition of startle in the rat: current knowledge and future challenges. Psychopharmacology 2001; 156: 194-215. http://dx.doi.org/10.1007/s002130100799 PMID 11549223
- ↑ Paylor R, Crawley JN. (1997) Inbred strain differences in prepulse inhibition of the mouse startle response. Psychopharmacology 1997; 132: 169-180. http://dx.doi.org/10.1007/s002130050333 PMID 9266614
- ↑ Aasen I, Kolli L, Kumari V. Sex effects in prepulse inhibition and facilitation of the acoustic startle response: implications for pharmacological and treatment studies. J Psychopharmacol. 2005 Jan;19(1):39-45. PMID 15671127
- ↑ Hoffman HS, Stitt CL. Inhibition of the glabella reflex by monaural and binaural stimulation. J Exp Psychol Hum Percept Perform. 1980 Nov;6(4):769-76. PMID 6449543
- ↑ Kumari V, Fannon D, Sumich AL, Sharma T. (2007) Startle gating in antipsychotic-naive first episode schizophrenia patients: One ear is better than two. Psychiatry Res. 2007 Mar 21; [Epub ahead of print] PMID 17382404
- ↑ Amsterdam; New York : Elsevier, 2001 Attraction, Distraction and Action: multiple perspectives on attentional capture; By Charles L. Folk, Bradley S. Gibson. ISBN 0444506764 Google books
- ↑ Filion DL, Dawson ME, Schell AM. (1993) Modification of the acoustic startle-reflex eyeblink: a tool for investigating early and late attentional processes. Biol Psychol. 1993 Jul;35(3):185-200. PMID 8218613
- ↑ Hoffman, H., Fleshler, M. (1963, September 6). Startle reaction: Modificationby background acoustic stimulation. Science, 141, 928–930. PMID 14043340
- ↑ Cadenhead KS, Geyer MA, Braff DL. Impaired startle prepulse inhibition and habituation in patients with schizotypal personality disorder. Am J Psychiatry. 1993 Dec;150(12):1862-7 PMID 8238643
- ↑ Swerdlow NR, Paulsen J, Braff DL, Butters N, Geyer MA, Swenson MR. Impaired prepulse inhibition of acoustic and tactile startle response in patients with Huntington's Disease. J Neurol Neurosur Psychiatry 1995; 58: 192-200. PMID 7876851
- ↑ Morton, N., Gray, N.S., Mellers, J., Toone, B., Lishman, W.A., & Gray, J.A. (1994). Prepulse inhibition in temporal lobe epilepsy. Schizophrenic Research, 15, 191.
- ↑ Braff D, Stone C, Callaway E, Geyer M, Glick I, Bali L. Prestimulus effects on human startle reflex in normals and schizophrenics. Psychophysiology. 1978 Jul;15(4):339-43. PMID 693742
- ↑ Kumari V, Das M, Zachariah E, Ettinger U, Sharma T. Reduced prepulse inhibition in unaffected siblings of schizophrenia patients.Psychophysiology. 2005 Sep;42(5):588-94. PMID 16176381
- ↑ Cadenhead KS, Swerdlow NR, Shafer KM, Diaz M, Braff DL. Modulation of the startle response and startle laterality in relatives of schizophrenic patients and in subjects with schizotypal personality disorder: evidence of inhibitory deficits. Am J Psychiatry. 2000 Oct;157(10):1660-8. Erratum in: Am J Psychiatry 2000 Nov;157(11):1904. PMID 11007721
- ↑ Hazlett EA, Romero MJ, Haznedar MM, New AS, Goldstein KE, Newmark RE, Siever LJ, Buchsbaum MS. (2007) Deficient attentional modulation of startle eyeblink is associated with symptom severity in the schizophrenia spectrum. Schizophr Res. 2007 May 1; PMID 17478083
- ↑ Mansbach RS, Geyer MA, Braff DL. (1988) Dopaminergic stimulation disrupts sensorimotor gating in the rat. Psychopharmacology 1988; 94: 507-514. PMID 3131796
- ↑ Swerdlow NR, Keith VA, Braff DL, Geyer MA. (1991) Effects of spiperone, raclopride, SCH 23390 and clozapine on apomorphine inhibition of sensorimotor gating of the startle response in the rat. J Pharmacol Exp Ther 1991; 256: 530-536. PMID 1825226
- ↑ 21.0 21.1 Swerdlow NR, Light GA, Cadenhead KS, Sprock J, Hsieh MH, Braff DL. (2006) Startle gating deficits in a large cohort of patients with schizophrenia. Arch Gen Psych. December, 2006;63:1325-1335. PMID 17146007
- ↑ Kumari V, Soni W, Sharma T. Influence of cigarette smoking on prepulse inhibition of the acoustic startle response in schizophrenia. Hum Psychopharmacol. 2001 Jun;16(4):321-326. PMID 12404567
- ↑ Leonard S, Adler LE, Benhammou K, Berger R, Breese CR, Drebing C, Gault J, Lee MJ, Logel J, Olincy A, Ross RG, Stevens K, Sullivan B, Vianzon R, Virnich DE, Waldo M, Walton K, Freedman R. Smoking and mental illness. Pharmacol Biochem Behav 2001;70:561-70
- ↑ De Leon J, Tracy J, McCann E, Mcgrory A, Diaz F. Schizophrenia and tobacco smoking: a replication study in another US psychiatric hospital. Schizophr Res 2002;56:55-65
- ↑ Gene Overview of All Published Schizophrenia-Association Studies for CHRFAM7A – Schizophrenia Gene Database.
- ↑ Gene Overview of All Published Schizophrenia-Association Studies for CHRNA7 – Schizophrenia Gene Database
- ↑ Paylor R, Nguyen M, Crawley JN, Patrick J, Beaudet A, Orr-Urtreger A. Alpha7 nicotinic receptor subunits are not necessary for hippocampal-dependent learning or sensorimotor gating: a behavioral characterization of alpha7-deficient mice. Learn Mem 1998; 5: 302-316
- ↑ Gogos JA, Morgan M, Luine V, Santha M, Ogawa S, Pfaff D et al. Catechol-O-methyltransferase-deficient mice exhibit sexually dimorphic changes in catecholamine levels and behavior. Proc Natl Acad Sci 1998; 95: 9991-9996. PMID 9707588 free fulltext
- ↑ Swerdlow NR, Geyer MA. (1998) Using an animal model of deficient sensorimotor gating to study the pathophysiology and new treatments of schizophrenia. Schizophr Bull 1998; 24:285-301 PMID 9613626 free fulltext
- ↑ Geyer, MA, Krebs-Thomson, K, Braff, DL, Swerdlow, NR. Pharmacological studies of prepulse inhibition models of sensorimotor gating deficits in schizophrenia: a decade in review. Psychopharmacology 2001; 156:117–154. PMID 11549216
- ↑ Light GA, Braff DL. (2001) Measuring P50 suppression and prepulse inhibition in a single recording session. Am J Psychiatry. 2001 Dec;158(12):2066-8. PMID 11729028 free full text
ru:Преимпульсное ингибирование
| This page uses Creative Commons Licensed content from Wikipedia (view authors). |