Assessment |
Biopsychology |
Comparative |
Cognitive |
Developmental |
Language |
Individual differences |
Personality |
Philosophy |
Social |
Methods |
Statistics |
Clinical |
Educational |
Industrial |
Professional items |
World psychology |
Biological: Behavioural genetics · Evolutionary psychology · Neuroanatomy · Neurochemistry · Neuroendocrinology · Neuroscience · Psychoneuroimmunology · Physiological Psychology · Psychopharmacology (Index, Outline)
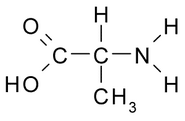
In chemistry, an amino acid is any molecule that contains both amine and carboxylic acid functional groups. In biochemistry, this shorter and more general term is frequently used to refer to alpha amino acids: those amino acids in which the amino and carboxylate functionalities are attached to the same carbon, the so-called α–carbon.
An amino acid residue is what is left of an amino acid once a molecule of water has been lost (an H+ from the nitrogenous side and an OH- from the carboxylic side) in the formation of a peptide bond.
Overview[]
Amino acids are the basic structural building units of proteins. They form short polymer chains called peptides or polypeptides which in turn form structures called proteins. The process of such formation is known as translation, which is part of protein synthesis.
Phenylalanine is one of the standard amino acids.
Twenty amino acids are encoded by the standard genetic code and are called proteinogenic or standard amino acids. At least two others are also coded by DNA in a non-standard manner as follows:
- Selenocysteine is incorporated into some proteins at a UGA codon, which is normally a stop codon.
- Pyrrolysine is used by some methanogens in enzymes that they use to produce methane. It is coded for similarly to selenocysteine but with the codon UAG instead.
Other amino acids contained in proteins are usually formed by post-translational modification, which is modification after translation in protein synthesis. These modifications are often essential for the function of the protein.
Proline is the only proteinogenic amino acid whose side group is cyclic and links to the a-amino group, forming a secondary amino group. Formerly, proline was misleadingly called an imino acid.
Over one hundred amino acids have been found in nature. Some of these have been detected in meteorites, especially in a type known as carbonaceous chondrites. Microorganisms and plants often produce very uncommon amino acids, which can be found in peptidic antibiotics (e.g., nisin or alamethicin). Lanthionine is a sulfide-bridged alanine dimer which is found together with unsaturated amino acids in lantibiotics (antibiotic peptides of microbial origin). 1-Aminocyclopropane-1-carboxylic acid (ACC) is a small disubstituted cyclic amino acid and a key intermediate in the production of the plant hormone ethylene.
In addition to protein synthesis, amino acids have other biologically-important roles. Glycine and glutamate are neurotransmitters as well as standard amino acids in proteins. Many amino acids are used to synthesize other molecules, for example:
- tryptophan is a precursor of the neurotransmitter serotonin
- glycine is one of the reactants in the synthesis of porphyrins such as heme.
Numerous non-standard amino acids are also biologically-important: Gamma-aminobutyric acid is another neurotransmitter, carnitine is used in lipid transport within a cell, ornithine, citrulline, homocysteine, hydroxyproline, hydroxylysine, and sarcosine.
Some of the 20 standard amino acids are called essential amino acids because they cannot be synthesized by the body from other compounds through chemical reactions, but instead must be taken in with food. In humans, the essential amino acids are lysine, leucine, isoleucine, methionine, phenylalanine, threonine, tryptophan, valine. Histidine and arginine is generally considered only essential in children, because of their inability to synthesise it given their undeveloped metabolism.
The phrase "branched-chain amino acids" is sometimes used to refer to the aliphatic amino acids: leucine, isoleucine and valine.
General structure[]
The general structure of proteinogenic alpha amino acids is:
R
|
H2N-C-COOH
|
H
Where R represents a side chain specific to each amino acid. Amino acids are usually classified by the properties of the side chain into four groups. The side chain can make them behave like a weak acid, a weak base, a hydrophile, if they are polar, and hydrophobe if they are nonpolar.
Isomerism[]
Except for glycine, where R = H, amino acids occur in two possible optical isomers, called D and L. Using the newer Cahn Ingold Prelog priority rules for designating the configuration of optical isomers, the L isomer would assigned the letter S and the D isomer would be assigned the letter R. The L (or S) amino acids represent the vast majority of amino acids found in proteins. D (or R) amino acids are found in some proteins produced by exotic sea-dwelling organisms, such as cone snails. They are also abundant components of the cell walls of bacteria.
This information is partially incorrect. L and D isomers are not the same as S and R isomers. S or R isomers may rotate the plane of polarized light to the left or to the right.
Reactions[]
Proteins are created by polymerization of amino acids by peptide bonds in a process called translation. This condensation reaction yields the newly formed peptide bond and a molecule of water.
1. Amino acid; 2, zwitterion structure; 3, two amino acids forming a peptide bond. (See also bond.)
List of standard amino acids[]
Structures[]
Structures and symbols of the 20 amino acids present in genetic code.
Chemical properties[]
Following is a table listing the one letter symbols, the three-letter symbols, and the chemical properties of the side chains of the standard amino acids. The mass listed is the weighted average of all common isotopes, and includes the mass of H2O. The one-letter symbol for an undetermined amino acid is X. The three-letter symbol Asx or one-letter symbol B means the amino acid is either asparagine or aspartic acid, whereas Glx or Z means either glutamic acid or glutamine. The three-letter symbol Sec or one-letter symbol U refers to selenocysteine. The letters J and O are not used.
| Abbrev. | Full Name | Side chain type | Mass | pI | pK1 (α-COOH) |
pK2 (α-+NH3) |
pKr (R) | Remarks | |
|---|---|---|---|---|---|---|---|---|---|
| A | Ala | Alanine | hydrophobic | 89.09 | 6.01 | 2.35 | 9.87 | Very abundant, very versatile. More stiff than glycine, but small enough to pose only small steric limits for the protein conformation. It behaves fairly neutrally, can be located in both hydrophilic regions on the protein outside and the hydrophobic areas inside. | |
| C | Cys | Cysteine | hydrophobic (Nagano, 1999) | 121.16 | 5.05 | 1.92 | 10.70 | 8.18 | The sulfur atom binds readily to heavy metal ions. Under oxidizing conditions, two cysteines can join together by a disulfide bond to form the amino acid cystine. When cystines are part of a protein, insulin for example, this enforces tertiary structure and makes the protein more resistant to unfolding and denaturation; disulphide bridges are therefore common in proteins that have to function in harsh environments, digestive enzymes (e.g., pepsin and chymotrypsin), structural proteins (e.g., keratin), and proteins too small to hold their shape on their own (eg. insulin). |
| D | Asp | Aspartic acid | acidic | 133.10 | 2.85 | 1.99 | 9.90 | 3.90 | Behaves similarly to glutamic acid. Carries a hydrophilic acidic group with strong negative charge. Usually is located on the outer surface of the protein, making it water-soluble. Binds to positively-charged molecules and ions, often used in enzymes to fix the metal ion. When located inside of the protein, aspartate and glutamate are usually paired with arginine and lysine. |
| E | Glu | Glutamic acid | acidic | 147.13 | 3.15 | 2.10 | 9.47 | 4.07 | Behaves similar to aspartic acid. Has longer, slightly more flexible side chain. |
| F | Phe | Phenylalanine | hydrophobic | 165.19 | 5.49 | 2.20 | 9.31 | Essential for humans. Phenylalanine, tyrosine, and tryptophan contain large rigid aromatic group on the side chain. These are the biggest amino acids. Like isoleucine, leucine and valine, these are hydrophobic and tend to orient towards the interior of the folded protein molecule. | |
| G | Gly | Glycine | hydrophilic | 75.07 | 6.06 | 2.35 | 9.78 | Because of the two hydrogen atoms at the α carbon, glycine is not optically active. It is the tiniest amino acid, rotates easily, adds flexibility to the protein chain. It is able to fit into the tightest spaces, e.g., the triple helix of collagen. As too much flexibility is usually not desired, as a structural component it is less common than alanine. | |
| H | His | Histidine | basic | 155.16 | 7.60 | 1.80 | 9.33 | 6.04 | In even slightly acidic conditions protonation of the nitrogen occurs, changing the properties of histidine and the polypeptide as a whole. It is used by many proteins as a regulatory mechanism, changing the conformation and behavior of the polypeptide in acidic regions such as the late endosome or lysosome, enforcing conformation change in enzymes. However only a few histidines are needed for this, so it is comparatively scarce. |
| I | Ile | Isoleucine | hydrophobic | 131.17 | 6.05 | 2.32 | 9.76 | Essential for humans. Isoleucine, leucine and valine have large aliphatic hydrophobic side chains. Their molecules are rigid, and their mutual hydrophobic interactions are important for the correct folding of proteins, as these chains tend to be located inside of the protein molecule. | |
| K | Lys | Lysine | basic | 146.19 | 9.60 | 2.16 | 9.06 | 10.54 | Essential for humans. Behaves similarly to arginine. Contains a long flexible side-chain with a positively-charged end. The flexibility of the chain makes lysine and arginine suitable for binding to molecules with many negative charges on their surfaces. E.g., DNA-binding proteins have their active regions rich with arginine and lysine. The strong charge makes these two amino acids prone to be located on the outer hydrophilic surfaces of the proteins; when they are found inside, they are usually paired with a corresponding negatively-charged amino acid, e.g., aspartate or glutamate. |
| L | Leu | Leucine | hydrophobic | 131.17 | 6.01 | 2.33 | 9.74 | Essential for humans. Behaves similar to isoleucine and valine. See isoleucine. | |
| M | Met | Methionine | hydrophobic | 149.21 | 5.74 | 2.13 | 9.28 | Essential for humans. Always the first amino acid to be incorporated into a protein; sometimes removed after translation. Like cysteine, contains sulfur, but with a methyl group instead of hydrogen. This methyl group can be activated, and is used in many reactions where a new carbon atom is being added to another molecule. | |
| N | Asn | Asparagine | hydrophilic | 132.12 | 5.41 | 2.14 | 8.72 | Neutralized version of aspartic acid. | |
| P | Pro | Proline | hydrophobic | 115.13 | 6.30 | 1.95 | 10.64 | Contains an unusual ring to the N-end amine group, which forces the CO-NH amide sequence into a fixed conformation. Can disrupt protein folding structures like α helix or β sheet, forcing the desired kink in the protein chain. Common in collagen, where it undergoes a posttranslational modification to hydroxyproline. Uncommon elsewhere. | |
| Q | Gln | Glutamine | hydrophilic | 146.15 | 5.65 | 2.17 | 9.13 | Neutralized version of glutamic acid. Used in proteins and as a storage for ammonia. | |
| R | Arg | Arginine | basic | 174.20 | 10.76 | 1.82 | 8.99 | 12.48 | Functionally similar to lysine. |
| S | Ser | Serine | hydrophilic | 105.09 | 5.68 | 2.19 | 9.21 | Serine and threonine have a short group ended with a hydroxyl group. Its hydrogen is easy to remove, so serine and threonine often act as hydrogen donors in enzymes. Both are very hydrophylic, therefore the outer regions of soluble proteins tend to be rich with them. | |
| T | Thr | Threonine | hydrophilic | 119.12 | 5.60 | 2.09 | 9.10 | Essential for humans. Behaves similarly to serine. | |
| V | Val | Valine | hydrophobic | 117.15 | 6.00 | 2.39 | 9.74 | Essential for humans. Behaves similarly to isoleucine and leucine. See isoleucine. | |
| W | Trp | Tryptophan | hydrophobic | 204.23 | 5.89 | 2.46 | 9.41 | Essential for humans. Behaves similarly to phenylalanine and tyrosine (see phenylalanine). Precursor of serotonin. | |
| Y | Tyr | Tyrosine | hydrophobic | 181.19 | 5.64 | 2.20 | 9.21 | 10.46 | Behaves similarly to phenylalanine and tryptophan (see phenylalanine). Precursor of melanin, epinephrine, and thyroid hormones. |
| Amino acid | Abbrev. | Side chain | Hydro- phobic | Polar | Charged | Small | Tiny | Aromatic or Aliphatic | van der Waals volume | Codon | Occurrence in proteins (%) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Alanine | Ala, A | -CH3 | X | - | - | X | X | - | 67 | GCU, GCC, GCA, GCG | 7.8 |
| Cysteine | Cys, C | -CH2SH | X | - | - | X | - | - | 86 | UGU, UGC | 1.9 |
| Aspartate | Asp, D | -CH2COOH | - | X | negative | X | - | - | 91 | GAU, GAC | 5.3 |
| Glutamate | Glu, E | -CH2CH2COOH | - | X | negative | - | - | - | 109 | GAA, GAG | 6.3 |
| Phenylalanine | Phe, F | -CH2C6H5 | X | - | - | - | - | Aromatic | 135 | UUU, UUC | 3.9 |
| Glycine | Gly, G | -H | X | - | - | X | X | - | 48 | GGU, GGC, GGA, GGG | 7.2 |
| Histidine | His, H | -CH2-C3H3N2 | - | X | positive | - | - | Aromatic | 118 | CAU, CAC | 2.3 |
| Isoleucine | Ile, I | -CH(CH3)CH2CH3 | X | - | - | - | - | Aliphatic | 124 | AUU, AUC, AUA | 5.3 |
| Lysine | Lys, K | -(CH2)4NH2 | - | X | positive | - | - | - | 135 | AAA, AAG | 5.9 |
| Leucine | Leu, L | -CH2CH(CH3)2 | X | - | - | - | - | Aliphatic | 124 | UUA, UUG, CUU, CUC, CUA, CUG | 9.1 |
| Methionine | Met, M | -CH2CH2SCH3 | X | - | - | - | - | - | 124 | AUG | 2.3 |
| Asparagine | Asn, N | -CH2CONH2 | - | X | - | X | - | - | 96 | AAU, AAC | 4.3 |
| Proline | Pro, P | -CH2CH2CH2- | X | - | - | X | - | - | 90 | CCU, CCC, CCA, CCG | 5.2 |
| Glutamine | Gln, Q | -CH2CH2CONH2 | - | X | - | - | - | - | 114 | CAA, CAG | 4.2 |
| Arginine | Arg, R | -(CH2)3NH-C(NH)NH2 | - | X | positive | - | - | - | 148 | CGU, CGC, CGA, CGG, AGA, AGG | 5.1 |
| Serine | Ser, S | -CH2OH | - | X | - | X | X | - | 73 | UCU, UCC, UCA, UCG, AGU,AGC | 6.8 |
| Threonine | Thr, T | -CH(OH)CH3 | X | X | - | X | - | - | 93 | ACU, ACC, ACA, ACG | 5.9 |
| Valine | Val, V | -CH(CH3)2 | X | - | - | X | - | Aliphatic | 105 | GUU, GUC, GUA, GUG | 6.6 |
| Tryptophan | Trp, W | -CH2C8H6N | X | - | - | - | - | Aromatic | 163 | UGG | 1.4 |
| Tyrosine | Tyr, Y | -CH2-C6H4OH | X | X | - | - | - | Aromatic | 141 | UAU, UAC | 3.2 |
Hydrophilic and hydrophobic amino acids[]
Depending on how polar the side chain, aminoacids can be hydrophilic or hydrophobic to various degree. This influences their interaction with other structures, both within the protein itself and within other proteins. The distribution of hydrophilic and hydrophobic aminoacids determines the tertiary structure of the protein, and their physical location on the outside structure of the proteins influences their quaternary structure. For example, soluble proteins have surfaces rich with polar aminoacids like serine and threonine, while integral membrane proteins tend to have outer ring of hydrophobic aminoacids that anchors them to the lipid bilayer, and proteins anchored to the membrane have a hydrophobic end that locks into the membrane. Similarly, proteins that have to bind to positive-charged molecules have surfaces rich with negatively charged aminoacids like glutamate and aspartate, while proteins binding to negative-charged molecules have surfaces rich with positively charged chains like lysine and arginine.
Hydrophilic and hydrophobic interactions of the proteins do not have to rely only on aminoacids themselves. By various posttranslational modifications other chains can be attached to the proteins, forming hydrophobic lipoproteins or hydrophylic glycoproteins.
Nonstandard amino acids[]
Aside from the twenty standard amino acids and the two special amino acids, selenocysteine and pyrrolysine, already mentioned above, there is a vast number of "nonstandard amino acids" which are not used in the body's regular manufacturing of proteins. Examples of nonstandard amino acids include the sulfur-containing taurine and the neurotransmitters GABA and dopamine. Other examples are lanthionine, 1-amino isobutyric acid, dehydroalanine, dehydro-amino-butyric acid,
Nonstandard amino acids are usually formed through modifications to standard amino acids. For example, taurine can be formed by the decarboxylation of cysteine, while dopamine is synthesized from tyrosine and hydroxyproline is made by a posttranslational modification from proline.
Uses of substances derived from amino acids[]
- Aspartame (aspartyl-phenylalanine-1-methyl ester) is an artificial sweetener.
- 5-HTP (5-hydroxytryptophan) has been used to treat neurological problems associated with PKU (phenylketonuria), as well as depression (as an alternative to L-Tryptophan).
- L-DOPA (L-dihydroxyphenylalanine) is a drug used to treat Parkinsonism.
- Monosodium glutamate is a food additive to enhance flavor.
See also[]
- Essential amino acids
- Strecker amino acid synthesis
References[]
- Doolittle, R.F. (1989) Redundancies in protein sequences. In Predictions of Protein Structure and the Principles of Protein Conformation (Fasman, G.D. ed) Plenum Press, New York, pp. 599-623
- David L. Nelson and Michael M. Cox, Lehninger Principles of Biochemistry, 3rd edition, 2000, Worth Publishers, ISBN 1572591536
- On the hydrophobic nature of cysteine.
External links[]
- Molecular Expressions: The Amino Acid Collection - Has detailed information and microscopy photographs of each amino acid.
- 22nd amino acid - Press release from Ohio State claiming discovery of a 22nd amino acid.
th:กรดอะมิโน bg:Аминокиселина ca:Aminoàcid cs:Aminokyselina da:Aminosyre de:Aminosäuren et:Aminohapped es:Aminoácido eo:Aminoacido fa:اسیدهای آمینه fr:Acide aminé gl:Aminoácido ko:아미노산 io:Amin-acido he:חומצת אמינו lt:Aminorūgštis lv:Aminoskābe lb:Aminosaier hu:Aminosav mk:Амино киселина nl:Aminozuur no:Aminosyre nn:Aminosyre pt:Aminoácido ru:Аминокислоты sl:Aminokislina sr:Аминокиселина su:Asam amino fi:Aminohappo sv:Aminosyra th:กรดอะมิโน uk:Амінокислота zh:氨基酸
| This page uses Creative Commons Licensed content from Wikipedia (view authors). |