Assessment |
Biopsychology |
Comparative |
Cognitive |
Developmental |
Language |
Individual differences |
Personality |
Philosophy |
Social |
Methods |
Statistics |
Clinical |
Educational |
Industrial |
Professional items |
World psychology |
Biological: Behavioural genetics · Evolutionary psychology · Neuroanatomy · Neurochemistry · Neuroendocrinology · Neuroscience · Psychoneuroimmunology · Physiological Psychology · Psychopharmacology (Index, Outline)
| 3β,17-dihydroxypregn-5-en-20-one IUPAC name | |
| CAS number 387-79-1 |
ATC code [[ATC_code_|]][2] |
| PubChem 3032570 |
DrugBank [3] |
| Chemical formula | {{{chemical_formula}}} |
| Molecular weight | 332.48 g/mol |
| Bioavailability | |
| Metabolism | AdrenalGonads |
| Elimination half-life | |
| Excretion | |
| Pregnancy category | |
| Legal status | |
| Routes of administration | |
17-Hydroxypregnenolone (also 17-OH-pregnenolone and 17α-hydroxypregnenolone), is a C21 steroid that is obtained by hydroxylation of pregnenolone at the C17α position. This step is performed by the mitochondrial cytochrome P450 enzyme 17α-hydroxylase (CYP17A1) that is present in the adrenal and gonads. Peak levels are reached in humans at the end of puberty and then decline.[1] High levels are also achieved during pregnancy.
Prohormone[]
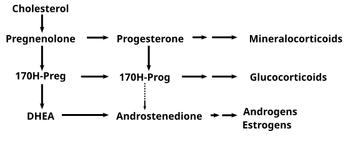
17 OH–pregnenolone is converted from pregnenolone and gives rise to DHEA (below) and to 17 OH-progesterone (to its right)
17-OH-pregnenolone is considered a prohormone in the formation of dehydroepiandrosterone (DHEA), itself a prohomone of the sex steroids.
This conversion is mediated by the enzyme 17,20 lyase . As such 17-OH-pregenolone represents an intermediary in the delta-5-pathway that leads from pregnenolone to DHEA. 17-hydroxypregneolone is also converted to 17-hydroxyprogesterone, a prohomone for glucocorticosteroids and androstenedione through the activity of 3-hydroxysteroid dehydrogenase.
Neurohormone[]
There is some evidence that 17-OH-pregnenolone may have activity as a neurohormone.[2]
Clinical use[]
Measurements of 17-OH-pregnenolone are useful in the diagnosis of certain forms of congenital adrenal hyperplasia.[3] In patients with congenital adrenal hyperplasia due to 3 beta-hydroxysteroid dehydrogenase deficiency 17-OH-pregnenolone is increased, while in patients with congenital adrenal hyperplasia due to 17 alpha-hydroxylase deficiency levels are low to absent.
See also[]
Congenital adrenal hyperplasia
Additional images[]
References[]
- ↑ Hill M, Lukac D, Lapcik O, Sulcova J, Hampl R, Pouzar V, Starka L. Age relationships and sex differences in serum levels of pregnenolone and 17-hydroxypregnenolone in healthy subjects. Clin Chem Lab Med. 1999 Apr;37(4):439-47. PMID 10369116
- ↑ Matsunaga M, Ukena K, Baulieu EE, Tsutsui K 7alpha-Hydroxypregnenolone acts as a neuronal activator to stimulate locomotor activity of breeding newts by means of the dopaminergic system. Proc Natl Acad Sci USA 2004 Dec 7;101(49):17282-7. PMID 15569930
- ↑ Riepe FG, Mahler P, Sippell, Partsch CJ. Longitudinal Study of Plasma Pregnenolone and 17-Hydroxypregnenolone in Full-Term and Preterm Neonates at Birth and during the Early Neonatal Period. The Journal of Clinical Endocrinology & Metabolism (2002) 87: 4301-4306 [1]
External links[]
| This page uses Creative Commons Licensed content from Wikipedia (view authors). |